 |
 |
- Search
| Arch Hand Microsurg > Volume 29(1); 2024 > Article |
|
Abstract
Purpose
A diaphyseal simple transverse fracture (DSTF) of the upper extremity (UE) requires direct anatomical reduction and absolute stability. No standard efficient method exists for reducing and maintaining a DSTF, despite its importance. Here, we introduce our transosseous wiring (TOW) method for UE-DSTFs.
Methods
To maintain reduction, the UE-DSTF was first fixed with TOW before definitive fixation with a locking plate across the fracture. We retrospectively reviewed 15 patients with at least 1 year of postoperative follow-up treatment from 2019 to 2021.
Results
All patients had achieved anatomical reduction and bone union at the final follow-up. Three patients had hardware removed because of irritation caused by the plate and screws. However, none of those three patients complained of irritation from the wire. One patient experienced refracture at the same site after hardware removal after a fall. The same technique was used in this case, and the bone union was observed 6 months after surgery.
For bone union, it is very important to decrease interfragmentary motion [1-4], which depends on the implant, its fixation in bone, and its inherent stability and rigidity [3].
Simple oblique fractures of the long bones in the upper extremity (UE) are treated by fixation with a lag screw and then a locking plate [1-3,5]. The plate-independent lag screw reduces the interfragmentary motion to achieve satisfactory bone healing and facilitate locking plate placement while maintaining the anatomical reduction of the fracture [1-3].
However, for transverse fractures in which the fracture plane is perpendicular to the long axis of the long bone, there is no fixation device like the lag screw used for oblique fractures. If there were a fixation device that played the same role as the fixation with a lag screw, it would be possible to control the interfragmentary motion, maintain anatomical reduction independently, and facilitate locking plate fixation.
We devised a surgical method using a steel wire for transverse long bone fractures that is similar to the lag screws used for oblique fractures. This study reports the clinical results of this method.
Ethics statement: This study was conducted after obtaining approval from the Institutional Review Board of Seoul National University College of Medicine (No. 2112-051-1281). Written informed consent was obtained from patients for the use of photographs in this study.
We retrospectively reviewed adults who underwent surgery for acute UE diaphyseal simple transverse fracture (DSTF) using our method treated from August 2019 to October 2021. All patients were treated by a trauma orthopedic surgeon with more than 15 years of experience. Only those with at least 1 year of postoperative follow-up were included. UE fractures include fractures of the clavicle, humerus, radius, and ulna. Complicated cases such as open fractures, concomitant osteomyelitis, atypical fractures, and pathological fractures were excluded. Pediatric patients with an open physis were also excluded. Fifteen patients (17 bones) were included, and we retrospectively reviewed their medical records and radiographic images of the fractures (Table 1, Fig. 1).
All patients underwent appropriate general or brachial plexus anesthesia and were prepared for routine surgical fracture fixation. After the fracture site was appropriately exposed, both ends of the fracture fragments were prepared for reduction and fixation. Minimal periosteal stripping and soft tissue dissection was performed so as not to compromise the biology of the bone. The bone cortices are called the “near cortex” and “far cortex” from the surgeon’s perspective. The fracture fragments were held with bone clamps and bicortical holes were drilled with a 2.5-mm drill bit 1 to 3 cm from the fracture site perpendicularly to the long axis, avoiding the position where the locking plate will be attached. Fig. 1 is a schematic illustration of this procedure. Two steel wires of appropriate thickness and length were prepared. Each wire was passed through the drill hole in each fragment. The two wires at the far cortex were twisted around each other 3 to 5 times so they would not become untied. After the fracture was reduced anatomically, the two wires at the near part were also twisted together. The space between the bones and the wire knot was reduced by further twisting until the desired tension was achieved. In a typical simple long bone fracture, the indentations at the fractured site will fit together even if the wire holes have imperfectly parallel positions. At this point, compression reduction of the DSTF was achieved and maintained by the two wires, facilitating the remaining surgical procedure. This construct could withstand the stress caused by maneuvers to place and fix the locking plate to protect the fracture construct, which is the function of a neutralization plate. The wires can be removed or left in place after complete fixation with the locking plate. In all our cases, after the twisted wires were trimmed to 5 mm in length and bent to give a low profile from the bone, no soft tissue irritation around the implant was observed.
Theoretically, a single wire can be used for this technique, but the passage at the far part may be hindered by the surrounding soft tissues and the far hole is often out of the surgeon’s field of view. Therefore, we thought that two wires would be a good way to facilitate wire passage through the bone holes.
If the desired position for the protecting plate is right on the wire knot, the plate can be positioned to the side because the wire knots can be brought into the plate holes. As the wire is pliable, screw placement close to the wire is usually possible. We used only a locking plate and screws for the definite fixation, with no additional device. The final fracture construct was inspected visually for macroscopic instability or interfragmentary motion before wound closure. Appropriate immobilization was applied depending on the location of the fracture. All immobilization was removed by 3 to 4 weeks postoperatively and all patients started normal daily activities and returned to normal physical exercise by 3 months postoperatively.
The patients were examined regularly as outpatients, including simple anteroposterior (AP), lateral, and oblique radiographs. After the bone union was observed, the size of the callus was measured, and the callus index was recorded.
Fracture union was defined by cortical continuity in both AP and lateral plain radiographs and no tenderness at the fracture site. When the plate made evaluation of the bone difficult in the AP and lateral views, an oblique view was obtained. Deformation, dislodgement, and implant failure were also assessed. Clinical findings such as signs of infection, soft tissue irritation, and other complications were also recorded.
The callus index was calculated by dividing the total callus diameter by the bone diameter (Fig. 2), using the radiographs with the lowest plate profile to measure the bone diameter clearly [6]. The callus index increases with callus growth, and a large index indicates greater interfragmentary motion at the fracture gap [7]. Because remodeling of the fracture callus does not start until the fracture is sufficiently stable, the maximum callus index is a useful indicator of the amount of interfragmentary motion during the healing period [8].
Bone union was achieved in all cases at an average of 6.4 weeks (range, 4–9 weeks). No case of infection, malunion, or metal failure was observed. Three patients complained of irritation and discomfort caused by a palpable implant, so the hardware was removed an average of 12 months (range, 6–13 months) postoperatively. In one of these patients, the same bone refractured 3 months after removing the hardware. This patient was treated using TOW with a longer locking plate and union was achieved 8 weeks postoperatively (case 7). The average callus index of all cases was 1.04 (range, 0.95–1.32).
For most UE-DSTF, anatomical reduction is easily achieved with an open procedure, but the challenge is maintaining the reduction until definitive fixation is achieved. There is no standard method for maintaining the reduction with interfragmentary compression of the transverse surfaces of the reduced fragments. Reduction clamps or forceps can be used, but they may hinder plate fixation when they are near the fixation site. If one chooses to apply the push–pull technique [9], it requires a longer skin incision for the additional screw and more soft tissue dissection. These conventional methods have multiple difficulties maneuvering, and unnecessary screw holes and cracks may lead to delayed union or iatrogenic fractures [10]. Therefore, we introduced a simple method of anatomical reduction and maintenance of UE-DSTF with interfragmentary compression until definitive fixation using transosseous wiring (TOW). A ‘transosseous wire technique’ is used in finger and facial bone fractures for fusing small joints or juxta-articular fractures [11-14]. To the best of our knowledge, this is the first report of TOW in UE-DSTF, including the clavicle.
TOW has been applied in the peri-articular areas of finger bones. Han et al [11]. applied TOW and intramedullary Kirschner wire fixation for arthrodesis of the distal interphalangeal joints in the hand. TOW has also been used in the osteosynthesis of the mandible [15]. These involved non-weight-bearing bones. Therefore, we adapted TOW to the long bones in the upper extremities. One has to differentiate TOW from a transosseous suture, which uses suture materials for enthesis repair. We used a steel wire to both approximate the fracture surfaces and maintain compression reduction until the final fixation. The working concept is similar to lag screw fixation in oblique fractures of the long bones that cause interfragmentary compression and guarantee reasonable stability, allowing plating [1-3]. TOW for a transverse fracture of a long bone is the same as lag screw fixation of an oblique fracture.
The advantage of this technique is that no additional special instrument is necessary, except a drill bit and steel wires. After this procedure, the normal bone is simply plated. TOW alone can provide interfragmentary compression, so the locking plate can function as a protection plate without an additional procedure to achieve interfragmentary compression, such as over-prebending or using a cortical screw in the dynamic compression hole. It provides additional stability to the plating construct. In this study, the final construct was achieved with locking plates protecting TOW [1-3]. Locking plates are stabilized with only locking screws for osteosynthesis. Anatomical reduction is achieved just through TOW, and the locking plates are modified to the bone’s shape using a bending device. Due to concerns that cortical bone screws might violate the integrity of the reduction, they were not employed. No excessive callus was observed during the union process, which means that the fracture healed with absolute stability during primary bone healing [7]. Therefore, TOW can be considered a “quasi-lag screw method” for simple transverse fractures. Because the plating can be done separately after the reduction is achieved with TOW, mini-open osteosynthesis is possible, which is our next study subject.
There were a few limitations to this study. First, a small number of patients with heterogeneous injuries was reviewed retrospectively. A cohort study with specific diagnoses may provide superior evidence. However, because this is the first report of a new surgical method, TOW in UE, we wanted to investigate the universal application of TOW, and the outcome was favorable. Specific studies may be required for each bone treated with our technique. Second, a biomechanical study of the TOW technique has not been reported. However, because the original fixation for UE-DSTF uses only a compression plate, TOW should increase the biomechanical stability. Third, the surgical time for the procedure was not measured because it was non-comparative consecutive case series, and the surgical situation was not controlled. However, we believe that TOW makes it easy to reduce and plate the fracture.
A noted complication of forearm shaft fractures is refracture following implant removal after bone union. This study also observed instances of refracture post-implant extraction. Potential causes include refracture due to premature removal of the implant when union is incomplete, and the possibility of refracture from structural weakening by stress shielding at the fracture site even after adequate union. Excessive compression from interosseous wiring could be another contributing factor. To mitigate this, the wire can be removed immediately after secure fixation of the fracture plate as a preventive strategy. The efficacy of this intervention could be validated through further research comparing refracture rates in patients with wires retained versus those removed after fixation.
Conflicts of interest
Min Bom Kim is a Deputy Editor of Archives of Hand and Microsurgery and was not involved in the review process of this article. All authors have no other conflicts of interest to declare.
Fig. 1.
Simple radiographs and gross image show various cases treated with our method. (A) A Galeazzi fracture shows a small butterfly fragment of a radial shaft fracture, which could be regarded as a simple transverse fracture. (B) A gross image shows the surgical field after the wiring between the fracture fragments. The radius is reduced anatomically with suboptimal stability, which can be tolerable for plate fixation. (C) After the application of transosseous wiring, the fracture is checked with intraoperative fluoroscopy. This provisional construct could be stable enough to be applied with the plate. (D) The final outcome of surgical treatment shows cortical bridging with minimal callus formation. The black arrows indicate the callus and bone diameters, which are measured for the callus index (callus diameter/bone diameter). (E) This image shows the application of this technique for a humerus shaft fracture. (F) These images show the clavicle shaft fracture using our method.
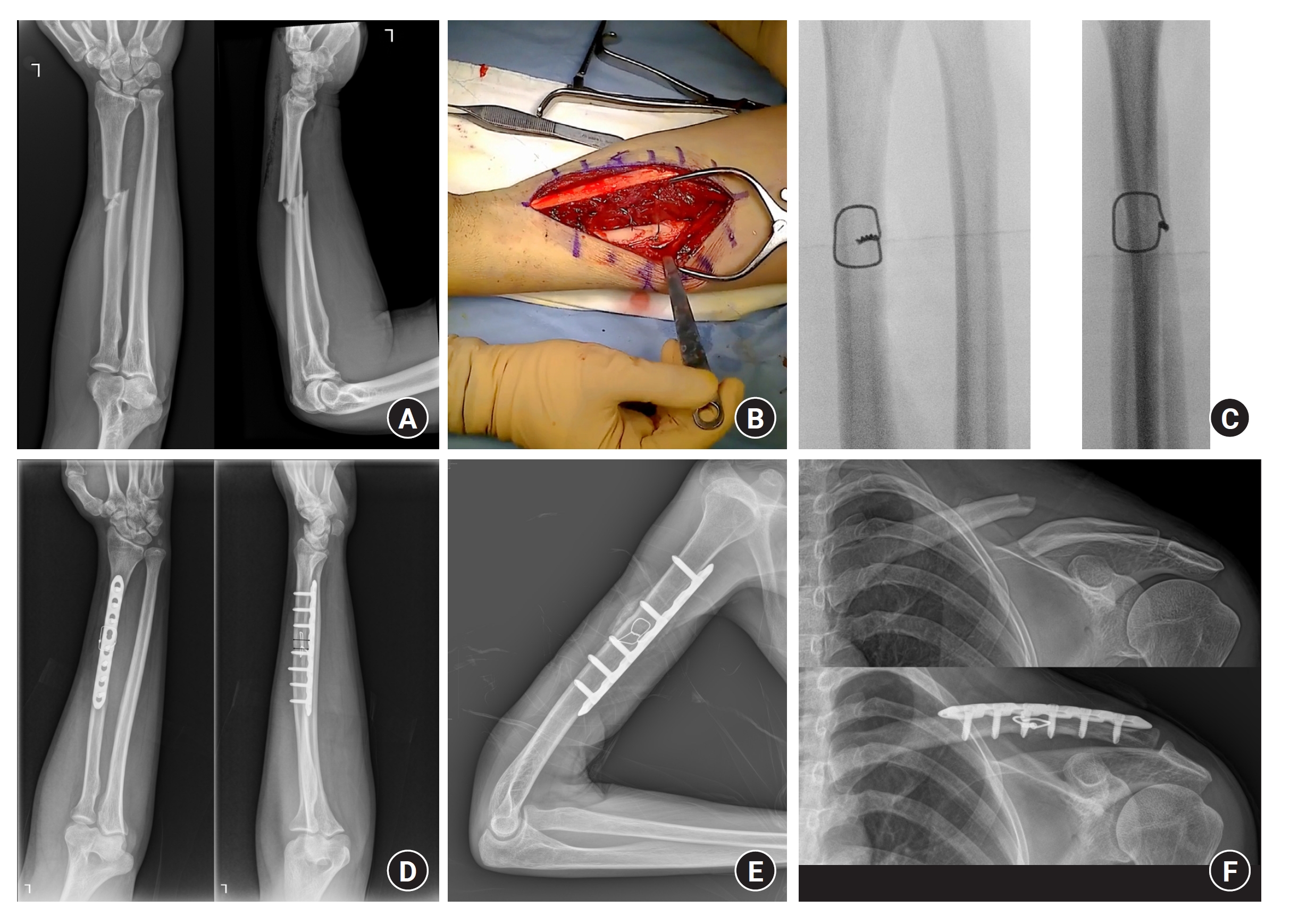
Fig. 2.
(A) A 23-year-old male patient had a radial shaft simple transverse fracture. (B) At 13 months postoperatively, fracture consolidation was confirmed. (C) The patient requested implant removal because of plate discomfort. (D) The fracture recurred 4 months after removal surgery at the same site as the initial fracture. (E) The same procedure was performed with transosseous wiring and longer locking plating, and 7 months after the second operation, the bone showed complete healing.
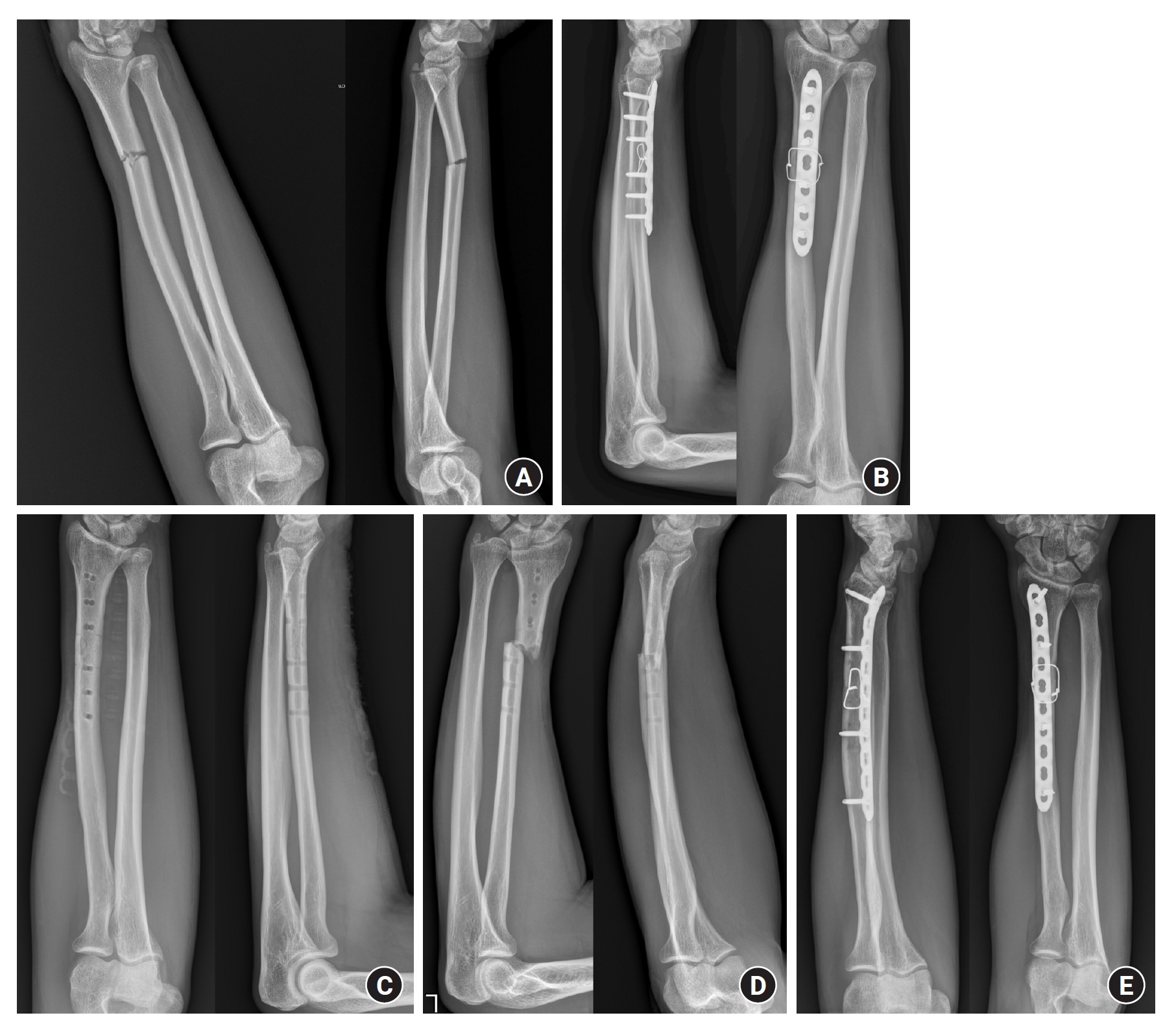
Table 1.
Patients’ demographics and clinical information
References
1. Märdian S, Schmölz W, Schaser KD, Duda GN, Heyland M. Interfragmentary lag screw fixation in locking plate constructs increases stiffness in simple fracture patterns. Clin Biomech (Bristol, Avon). 2015;30:814-9.


2. Märdian S, Schmölz W, Schaser KD, Duda GN, Heyland M. Locking plate constructs benefit from interfragmentary lag screw fixation with decreased shear movements and more predictable fracture gap motion in simple fracture patterns. Clin Biomech (Bristol, Avon). 2019;70:89-96.


3. Horn C, Döbele S, Vester H, Schäffler A, Lucke M, Stöckle U. Combination of interfragmentary screws and locking plates in distal meta-diaphyseal fractures of the tibia: a retrospective, single-centre pilot study. Injury. 2011;42:1031-7.


4. Leung F, Chow SP. Locking compression plate in the treatment of forearm fractures: a prospective study. J Orthop Surg (Hong Kong). 2006;14:291-4.



5. Plecko M, Lagerpusch N, Pegel B, Andermatt D, Frigg R, Koch R, et al. The influence of different osteosynthesis configurations with locking compression plates (LCP) on stability and fracture healing after an oblique 45° angle osteotomy. Injury. 2012;43:1041-51.


6. Gardner TN, Hardy J, Evans M, Kenwright J. Temporal changes in dynamic inter fragmentary motion and callus formation in fractures. J Biomech. 1997;30:315-21.


7. Aro HT, Chao EY. Bone-healing patterns affected by loading, fracture fragment stability, fracture type, and fracture site compression. Clin Orthop Relat Res. 1993;(293):8-17.

8. Eastaugh-Waring SJ, Joslin CC, Hardy JR, Cunningham JL. Quantification of fracture healing from radiographs using the maximum callus index. Clin Orthop Relat Res. 2009;467:1986-91.



9. Heim D, Luria S, Mosheiff R, Weil Y. Transverse simple fracture of the ulna [Internet]. In: Colton OL, editor. AO Surgery Reference. Davos: AO Foundation; 2013 [cited 2023 Feb 6]. Available from: https://surgeryreference.aofoundation.org/orthopedic-trauma/adult-trauma/forearm-shaft/transverse-simple-fracture-of-the-ulna/orif-compression-plating#reduction.
10. Court-Brwon CM, McQueen MM, McKee M, Tornetta P III. Techniques and instruments for fracture reduction. In: Rockwood CA, editor. Rockwood and Green's fractures in adults. 8th ed. Philadelphia: Wolters Kluwer Health; 2014. p. 206-14.
11. Han SH, Cha YS, Song WT. Arthrodesis of distal interphalangeal joints in the hand with interosseous wiring and intramedullary K-wire fixation. Clin Orthop Surg. 2014;6:401-4.



12. Githens T, Murphree J, Daneshfar C, MacKay B. Interosseus wiring of distal radius fractures with volar comminution. Tech Orthop. 2019;34:46-9.

13. Gingrass RP, Fehring B, Matloub H. Intraosseous wiring of complex hand fractures. Plast Reconstr Surg. 1980;66:383-94.









