 |
 |
- Search
| Arch Hand Microsurg > Volume 27(4); 2022 > Article |
|
Abstract
Purpose
This study conducted a comparative analysis of the effectiveness of split-thickness skin grafts (STSGs) and free flaps of the lateral thoracic region performed for coverage after extensive debridement in patients with difficult-to-treat chronic venous ulcers (CVUs) with severe symptoms.
Methods
This retrospective, single-center study included 20 patients (28 cases) with CVUs. Patients who received an STSG or free-flap procedure were included in the study. Data comparing these two groups were analyzed.
Results
The STSG and free-flap groups showed no significant differences in patient demographics. There was no significant difference in wound size before and after debridement between the two groups (before, 52.25±58.03 cm2 vs. 37.69±32.83 cm2, p=0.407; after, 210.92±202.80 cm2 vs. 142.63±84.01 cm2, p=0.291). Wound disruption was not significantly different between the groups (p=0.231). However, a significant difference was found in recurrence between the STSG group (n=7, 58.3%) and the free-flap group (n=1, 6.3%) (p=0.004).
Venous skin ulcer is the most severe form of chronic venous disease, which affects 1% to 3% of the general population and up to 4% of people aged ≥80 years [1,2]. The high recurrence rate of chronic venous ulcer (CVU) can cause significant physical and emotional distress to the patient, as well as high socioeconomic losses. Moreover, the incidence of CVU tends to increase with age and obesity, resulting in a gradual increase in social loss. It has been reported that 2.5 million to 3.0 million Americans are affected by venous insufficiency, with at least 600,000 patients suffering from chronic leg ulcers; as a result, the annual economic burden has reached up to $15 billion [3]. Although it is not a life-threatening disease, patients with CVU experience a significant decline in quality of life due to the high recurrence rate and severity of the symptoms. Despite this, a standardized treatment regime for CVU has not been established.
The key points for the treatment of CVU are wound management and lifelong compression therapy. Compression therapy is known to be an effective method for reducing edema and controlling venous hypertension, which has been identified as the cause of venous ulcers, and is actively used to treat venous ulcers [4,5]. Moreover, various biophysical therapies, including ultrasonography, electrostimulation, and phototherapy, have been used in recent years to treat CVU, while other methods, including stem cell therapy, plasma therapy, oxygen therapy, and antitumor necrosis factor therapy, have also been attempted. However, the clinical efficacy of these treatment modalities still needs to be proven and many of these methods are still in the experimental stage [6].
Surgical treatment is designed to promote the improvement of venous drainage and recovery of tissue damage, for which sclerotherapy, ligation or ablation of veins, venous valve reconstruction, and endoscopic perforator vein surgery may be considered. However, approximately one in five venous ulcer cases show no healing after 1 year despite such treatment [4]. For patients with repeated recurrence and severe open wounds, extensive debridement of pigmentation, eczema, and lipodermatosclerosis near the CVU may be unavoidable. Furthermore, a skin graft and tissue free-flap procedure may be performed on the defect that results from debridement.
The objective of the present study was to compare and assess the effectiveness of a split-thickness skin graft (STSG) and the free flap of lateral thoracic region procedure performed for coverage after extensive debridement at Hanyang University Medial Center in patients with a difficult-to-treat CVU with severe symptoms.
Ethics statement: The study was conducted with approval from the Institutional Review Board of Hanyang University College of Medicine (No. 2022-05-007). Written informed consent for publication of the clinical images was obtained from the patient.
The study was a retrospective study of patients with CVU who received surgical treatment in our institution between May 2013 and January 2022. The diagnosis of CVU was based on a clinical diagnosis; among patients with pain, fatigue, and heaviness in the legs, those with the severe form of open wounds with edema, hyperpigmentation, and eczema were diagnosed with CVU. The patients who received simple debridement and negative-pressure wound therapy were excluded from the study. Only patients who received STSG and free flap of lateral thoracic region procedures were included in the study; data comparing these two groups were analyzed.
Patient demographics and characteristics were obtained from electronic medical records. Sex, age, body mass index (BMI), defect size, and comorbidities (diabetes mellitus, hypertension, and rheumatoid arthritis) were included. The same patient receiving multiple rounds of STSG, patients who underwent a subsequent free-flap procedure after STSG, and the same patient receiving surgical treatment on other affected areas were all included in the study data as separate cases.
All surgical procedures were performed by three experienced surgeons as a one-stage operation with concurrent debridement and coverage. Debridement involved wide excision of at least 2 cm and up to 11 cm from the wound margin. The extent of the debridement was decided based on the clinical determination on tissue conditions such as pigmentation and lipodermatosclerosis near the wound; the focus was on sufficient excision more than the defect size to be covered after debridement. Debridement was performed up to the suprafascial layer, and if the ulcer was already deeper than that, excision was performed including the muscle layer depending on the wound state. During the procedure, visibly abnormal vessels were ligated. Two methods were used for coverage. For STSG, the skin was harvested from the thigh according to the defect size, and when necessary, meshed STSG or Matriderm (MedSkin Solutions Dr. Suwelack AG, Billerbeck, Germany) was used. For the free-flap procedure, we selected the lateral thoracic area as the donor site and mainly used the right side. The flap was harvested in various forms, such as a perforator, musculocutaneous, or chimeric flap, depending on the defect size and character. For the perforator type, the thoracodorsal artery perforator flap was used, and for the musculocutaneous type, the latissimus dorsi muscle was used. In addition, in the case of the chimeric type, the thoracodorsal artery perforator flap and the serratus anterior muscle flap were harvested together and used. As we mentioned above, since debridement was performed on the suprafascial layer, a perforator flap that can obtain a thin flap for aesthetic benefit after coverage was preferred. However, when an appropriate perforator could not be found or the depth of the defect was up to the muscle layer, a musculocutaneous flap type was used to acquire more sufficient volume. In the case of a chimeric-type flap, it was performed when a deep vessel was exposed after debridement or when a defect in some muscle layer remained after coverage using a perforator flap. For the recipient vessel, the anterior tibial or posterior tibial vessel was used depending on the location of the defect; it was determined by referencing preoperative computed tomography angiography results (Fig. 1).
The choice of surgical method was decided according to the surgeon’s judgment comprehensively considering clinical features including defect size and vessel patency. Procedures such as perforator ligation surgery, sclerotherapy, and valve surgery were not performed before surgery. And in both groups, compression therapy was started 2 weeks after surgery if there were no complications. Compression was initially performed using an elastic bandage and was replaced with compression garments when the wound was stabilized and maintained continuously. Recurrence was defined as a newly discovered skin ulcer in the covered area or surrounding area within 6 months after surgery, regardless of the surgical method.
Categorical data were expressed as a percentage and analyzed using the chi-square or Fisher exact test. Continuous variables were calculated as the mean and standard deviation for comparison with a two-sided t-test and Levene test of equal variances. In cases where the same patient had received multiple surgical treatments, the outcome of each surgical treatment was analyzed separately according to the surgical method. The statistics derived reflect the data at the time of surgery. A p-value of <0.05 was considered statistically significant. All analyses were performed using IBM SPSS Statistics ver. 25 (IBM Corp., Armonk, NY, USA).
A total of 20 patients were included in the study: seven received STSG, 13 received free-flap procedures, and four received both. A comparison between the STSG and free-flap groups showed no significant difference in sex (p=0.253). Furthermore, there was no significant difference in mean age between the STSG (46.5±18.45 years) and free-flap groups (52.0±16.30 years) (p=0.411). In addition, no significant differences in height (p=0.860) or BMI (p=0.302) were observed between the two groups. Measurement of ulcer size before debridement showed a larger mean defect size in the STSG group (52.25±58.03 cm2) than in the free-flap group (37.69±32.83 cm2); however, the difference was not significant (p=0.407) (Table 1).
There were 12 cases in total of STSG, of which Matriderm and meshed STSG were used in two each. The mean period for recurrence of venous ulcer after STSG was 5.14±6.72 months. Meanwhile, there were 16 free-flap procedures, in which all patients had a history of receiving surgical treatment before the flap procedure. There were 11 patients who had previously received STSG for skin ulcers, and there was one who received up to four rounds of skin graft due to continued recurrence after the surgery. The mean follow-up period after the free-flap procedure was 35.63±28.27 months. With respect to the flap type, a perforator and musculocutaneous flap were harvested in 10 and three cases, respectively, while a chimeric type of flap was harvested in three cases, in which the serratus anterior muscle was elevated together with the thoracodorsal artery perforator flap. The mean flap size was 152.00±91.83 cm2. Partial flap loss occurred in three cases; healing was achieved in all cases through dressing and revision under local anesthesia. Meanwhile, there were no cases of total flap loss (Table 2).
Measurement of ulcer size after surgical debridement also showed a larger mean defect size in the STSG group (210.92±202.80 cm2) than in the free-flap group (142.63±84.01 cm2), just as with before debridement, although the difference was not significant (p=0.291). Wound disruption after coverage was found in five and three cases, respectively, showing no significant difference between the groups (p=0.231). Wound disruption occurred in one of the two cases in which Matriderm was used with STS, and was healed by dressing. With respect to recurrence, there was a significant difference in recurrence between the STSG (n=7, 58.3%) and free flap groups (n=1, 6.3%) (p=0.004; Table 3).
For CVU, surgical treatment such as sclerotherapy, valve reconstruction, and perforator vein surgery could be considered for improvement of the venous system causing CVU. However, patients with the chronic venous disease who have more severe pain and itching or have an open wound (Clinical-Etiology-Anatomy-Pathophysiology classification C6) may need more aggressive treatment. The treatment could be designed to remove the venous system that is causing the problem by performing extensive debridement on the wound and surrounding tissues that have begun to deform, followed by coverage of the defect.
Bitsch et al. [7] performed STSG on chronic leg ulcer cases and analyzed the prognosis of traumatic, arterial, and venous ulcers; STSG performed on venous ulcer cases showed a recurrence rate of 14% after 1 year. However, other studies have shown that recurrence is more common after a skin graft for CVU. Turczynski and Tarpila [8] performed STSG in 51 cases of venous ulcer and reported a recurrence rate of 43%, with a mean period of 4 months until such recurrence. Moreover, Abisi et al. [9] reported a recurrence rate of 42% based on a follow-up of up to 4 years in 72 cases of venous ulcers. In the present study, the recurrence rate after STSG was 58.3%, which was relatively higher than that of previous studies, while long-term follow-up of at least 1 year showed recurrence in all patients. Such findings indicated that it would be difficult to resolve venous hypertension, which is the fundamental cause of CVU, by debridement and coverage with simple tissue-like skin. STSG may be an option for incurable CVUs, but considering the pathophysiology of venous ulcers, a long-term therapeutic effect may not be expected, and as a result, there may be frequent recurrences.
Meanwhile, coverage by a free flap has been reported in various previous studies. Kumins et al. [10] reported no recurrence based on a long-term follow-up after a free-flap procedure using the rectus abdominis or latissimus dorsi in 24 cases of non-healing venous ulcer, including recurrences after STSG. Isenberg [11] treated CVU using a radial forearm fasciocutaneous free flap and reported satisfactory outcomes in short- and long-term follow-ups. In the present study, treatment of venous ulcers using a free flap showed a recurrence rate of 6.3%, which was significantly lower than that of 58.3% after STSG. It is believed that this could be due to the free flap resolving venous stasis caused by the dysfunctional venous valve, which has been identified as a cause of venous ulcers. Healthy tissue harvested for the free-flap procedure contains many normal valves, which can store and drain venous blood by replacing abnormal tissue [10]. While sclerotherapy [12], valvuloplasty [13], and embolization/ligation of veins, perforator vessels, and feeding vessels [14] are being attempted as surgical treatment for venous insufficiency, these are procedures performed on localized veins and are methods that simply “repair” the abnormal vein system. On the other hand, free-flap surgery that can “replace” many healthy venous systems can theoretically be a fundamental treatment modality for venous ulcers. Healthy tissue grafted in this manner can block the hemodynamic vicious cycle by resolving congestion in the existing lesion, which can affect tissues surrounding the flap [15]. In addition, highly vascularized free-flap tissue can achieve an improvement in abnormal vessels within several months after the surgery, and as a result, the venous system beyond the flap and venous insufficiency can be improved to lower the likelihood of recurrence. In this sense, the free-flap procedure could be a good option for the surgical treatment of venous ulcers.
Previous studies have pointed out that recurrence, especially a newly formed ulcer from the flap margin, is caused by insufficient debridement of lipodermatosclerotic tissue near the ulcer that may have appeared as normal skin during the previous surgery [4,11,16]. Theoretically, sufficient debridement could be a solution to this problem. In the surgical treatment of venous ulcers, the removal of many abnormal vessels and tissues by extensive debridement around the lesion is essential for a better prognosis. However, since coverage must also be considered, the extent of debridement is limited. Therefore, when considering coverage using free flaps, this is an important point in donor site selection.
In 1979, Watson et al. [17] became the first to attempt microvascular transfer of a latissimus dorsi muscle flap on the lower extremity. Different types of flaps, including the anterolateral thigh, superficial circumflex iliac artery perforator, gracilis, radial forearm, and rectus abdominis flap, are used to reconstruct the lower extremities [18-21]; however, the free tissue transfer of the lateral thoracic region could be preferred more by patients with a venous ulcer of lower extremities.
There are several advantages to treating CVU by selecting the lateral thoracic region as the donor site. First, as described above, extensive debridement is essential in the surgical treatment of venous ulcers, and so a tissue that can cover a wide range is required. The lateral thoracic region for free-flap tissue can be viewed as an appropriate choice since a large flap can be obtained. In the case of thoracodorsal artery perforator flaps, a skin paddle sized up to 12×25 cm can be harvested [22]. Ciudad et al. [23] reported clinically satisfactory outcomes by harvesting a flap 15×30 cm in size using the extended fleur-de-lis method. Second, in venous ulcer cases with exposed tendon, muscle, or bone after extensive debridement, a sufficient width and volume of the flap must be assured. Thus, there is the advantage of elevating various forms of flaps together, with a musculocutaneous flap and a chimeric flap harvested from the serratus anterior muscle. Mahajan et al. [24] reported the successful reconstruction of 47 traumatic extensive lower extremity wounds with latissimus dorsi-serratus anterior chimeric flaps. Tachi et al. [25] covered complex soft tissue and bone defects after osteomyelitis with a chimeric-type flap using latissimus dorsi myocutaneous flap and scapula bone. As in previous studies, in the present study, coverage was performed by harvesting different types of flaps depending on the defect, from which favorable treatment outcomes were achieved. In addition, the outcomes were aesthetically satisfactory.
The risk of recurrence exists from a long-term perspective, and skin grafts are still widely used as surgical treatment for CVU [26,27]. On the other hand, surgical treatment using the free flap has the disadvantage of requiring microsurgery, although it offers the major advantage of fundamentally resolving venous insufficiency. Ultimately, free tissue transfer has an advantage in that it can reduce the recurrence rate of incurable CVU, which is a problem due to frequent recurrence after treatment. The significance of the present study can be found in the fact that it directly compared the two surgical treatment methods most widely used for CVU, besides perforator surgery or venous ablation that focus on venous manipulation. However, the present study was a single-center retrospective study; thus, there are limitations in generalizing the findings. Because of such reasons, there may be selection bias when selecting STSG or free-flap procedures for treating CVU. In patients with a wider ulcer, STSG may have been preferred over a free flap due to the burden of coverage. However, despite the differences in the wound and defect sizes, there was no significant difference between the two groups. Moreover, because cases involving the same patient receiving multiple rounds of STSG or patients who received STSG subsequently receiving the free-flap procedure were all included as individual cases, each case may lack independence.
Free-flap surgery may be a good option for difficult-to-treat, recurrent CVU. Based on the nature of venous ulcers that require extensive debridement, a free flap from the lateral thoracic region that enables the harvesting of large and various forms of flaps could be the best choice for microsurgery.
Fig. 1.
A 27-year-old male patient with a chronic venous ulcer in the left lower leg previously received split-thickness skin grafts twice but relapsed. He complained of severe pain and recurring open wounds, which caused a detrimental impact on his daily life. (A) A skin ulcer with necrotic tissue, pigmentation of the surrounding soft tissue, and lipodermatosclerosis is observed. (B) After extensive debridement of the skin ulcer and surrounding tissue, coverage was performed using a thoracodorsal artery perforator flap. The posterior tibial vessel was used as the recipient vessel. (C) A photograph taken 9 months after surgery. No recurrence was observed, and the previous symptoms had all disappeared. In addition, the degeneration of the surrounding skin was resolved, providing satisfactory aesthetic results.
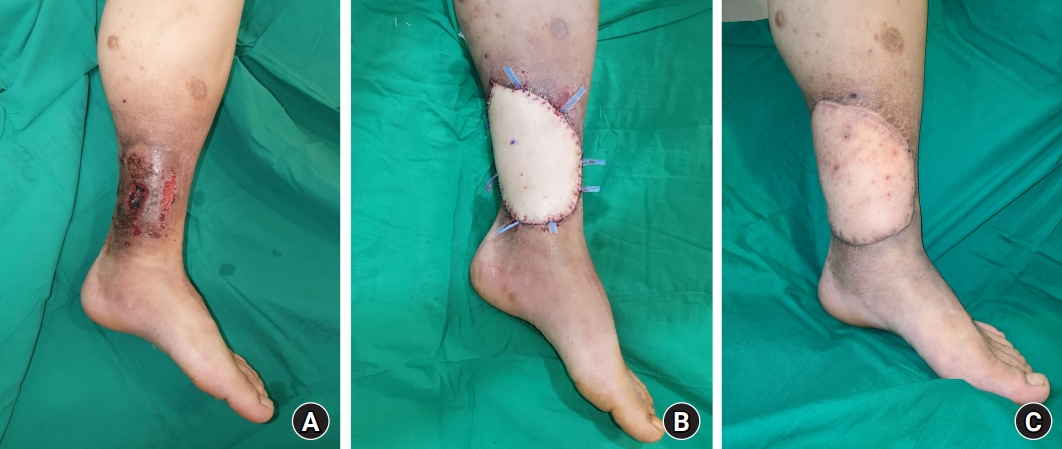
Table 1.
The patients’ characteristics
References
1. Bonkemeyer Millan S, Gan R, Townsend PE. Venous ulcers: diagnosis and treatment. Am Fam Physician. 2019;100:298-305.

3. Gould LJ, Dosi G, Couch K, et al. Modalities to treat venous ulcers: compression, surgery, and bioengineered tissue. Plast Reconstr Surg. 2016;138(3 Suppl):199S-208S.


4. Song MS, Baldwin AJ, Wormald JC, Coleman C, Chan JK. Outcomes of free flap reconstruction for chronic venous ulceration in the lower limb: a systematic review. Ann Plast Surg. 2022;89:331-5.


5. Health Quality Ontario. Compression stockings for the prevention of venous leg ulcer recurrence: a health technology assessment. Ont Health Technol Assess Ser. 2019;19:1-86.
6. Aleksandrowicz H, Owczarczyk-Saczonek A, Placek W. Venous leg ulcers: advanced therapies and new technologies. Biomedicines. 2021;9:1569.



7. Bitsch M, Saunte DM, Lohmann M, Holstein PE, Jørgensen B, Gottrup F. Standardised method of surgical treatment of chronic leg ulcers. Scand J Plast Reconstr Surg Hand Surg. 2005;39:162-9.


8. Turczynski R, Tarpila E. Treatment of leg ulcers with split skin grafts: early and late results. Scand J Plast Reconstr Surg Hand Surg. 1999;33:301-5.


9. Abisi S, Tan J, Burnand KG. Excision and meshed skin grafting for leg ulcers resistant to compression therapy. Br J Surg. 2007;94:194-7.



10. Kumins NH, Weinzweig N, Schuler JJ. Free tissue transfer provides durable treatment for large nonhealing venous ulcers. J Vasc Surg. 2000;32:848-54.


11. Isenberg JS. Additional follow-up with microvascular transfer in the treatment of chronic venous stasis ulcers. J Reconstr Microsurg. 2001;17:603-5.


12. Pihlaja T, Torro P, Ohtonen P, Romsi P, Pokela M. Ten years of experience with first-visit foam sclerotherapy to initiate venous ulcer healing. J Vasc Surg Venous Lymphat Disord. 2021;9:954-60.


13. Us MH, Ugur M. Has external banding become a historical technique during venous valve repair? Rev Assoc Med Bras (1992). 2021;67:1676-80.


15. Tark KC, Chung S. Histologic change of arteriovenous malformations of the face and scalp after free flap transfer. Plast Reconstr Surg. 2000;106:87-93.


16. Steffe TJ, Caffee HH. Long-term results following free tissue transfer for venous stasis ulcers. Ann Plast Surg. 1998;41:131-9.


17. Watson JS, Craig RD, Orton CI. The free latissimus dorsi myocutaneous flap. Plast Reconstr Surg. 1979;64:299-305.


18. Kozusko SD, Liu X, Riccio CA, et al. Selecting a free flap for soft tissue coverage in lower extremity reconstruction. Injury. 2019;50 Suppl 5:S32-9.


19. Irthum C, Fossat S, Bey E, Duhamel P, Braye F, Mojallal A. [Anterolateral thigh flap for distal lower leg reconstruction]. Ann Chir Plast Esthet. 2017 62:224-31. French.


20. Yamamoto T, Saito T, Ishiura R, Iida T. Quadruple-component superficial circumflex iliac artery perforator (SCIP) flap: a chimeric SCIP flap for complex ankle reconstruction of an exposed artificial joint after total ankle arthroplasty. J Plast Reconstr Aesthet Surg. 2016;69:1260-5.


21. Vranckx JJ, Misselyn D, Fabre G, Verhelle N, Heymans O, Van den hof B. The gracilis free muscle flap is more than just a “graceful” flap for lower-leg reconstruction. J Reconstr Microsurg. 2004;20:143-8.


22. Guerra AB, Metzinger SE, Lund KM, Cooper MM, Allen RJ, Dupin CL. The thoracodorsal artery perforator flap: clinical experience and anatomic study with emphasis on harvest techniques. Plast Reconstr Surg. 2004;114:32-43.


23. Ciudad P, Manrique OJ, Bustos SS, et al. The modified extended fleur-de-lis latissimus dorsi flap for various complex multi-directional large soft and bone tissue reconstruction. Cureus. 2020;12:e6974.



24. Mahajan RK, Srinivasan K, Bhamre A, Singh M, Kumar P, Tambotra A. A retrospective analysis of latissimus dorsi-serratus anterior chimeric flap reconstruction in 47 patients with extensive lower extremity trauma. Indian J Plast Surg. 2018;51:24-32.



25. Tachi M, Toriyabe S, Imai Y, Takeda A, Hirabayashi S, Sekiguchi J. Versatility of chimeric flap based on thoracodorsal vessels incorporating vascularized scapular bone and latissimus dorsi myocutaneous flap in reconstructing lower-extremity bone defects due to osteomyelitis. J Reconstr Microsurg. 2010;26:417-24.









