 |
 |
- Search
| Arch Hand Microsurg > Volume 27(3); 2022 > Article |
|
Abstract
Purpose
Free flaps play an important role in the reconstruction of chronic wounds in the lower extremities. Nonetheless, the use of free flaps for chronic wound reconstruction tends to be challenging. A considerable number of patients with chronic wounds have noteworthy medical histories, such as chronic kidney disease or end-stage renal disease (ESRD). This study aimed to determine the relationship between the success rate and complications of free flap surgery according to the degree of kidney disease from normal renal function to ESRD.
Methods
A retrospective study of all patients who underwent free tissue transfer procedures due to chronic wounds in the lower-extremity area over a 9-year period, between October 2011 and February 2020, was conducted. The primary outcome was flap failure, and major complications were set as the secondary outcome.
Results
Sixty-seven consecutive free flaps were used for chronic wounds of the lower extremities at our institution. Flap failure occurred in six patients (9.0%), resulting in a total flap survival rate of 91.0%. Sixteen patients (23.9%) experienced major complications. Flap failure was correlated with the presence of ESRD and renal transplantation.
The free flap plays an important role in the reconstruction of chronic wounds in the lower extremities. Reconstruction by free flap can be performed even when white structures such as bones or tendons are exposed, and it can be a better choice than a skin graft in weight-bearing or joint areas [1].
Nonetheless, the use of free flaps for chronic wound reconstruction tends to be challenging in most cases [2,3]. A considerable number of chronic wound patients have varying medical histories, such as ischemic heart disease, peripheral vascular disease (PVD), history of cerebrovascular attack, chronic kidney disease (CKD), or end-stage renal disease (ESRD) [4]. Free flap placement in patients with comorbid illnesses is difficult, and the rate of flap complications is relatively high. In particular, CKD and ESRD were considered as risk factors for free flap in some studies [5-10].
In this study, patients who underwent free flap surgery for chronic wounds of the lower extremities were examined, and their relationship with the success rate and complications of free flap surgery according to the degree of their kidney disease from normal renal function to ESRD was investigated.
Ethics statement: The study was conducted after obtaining approval from the Institutional Review Board of Ajou University Hospital (No. AJIRB-MED-MDB-20-374) and the research was conducted according to the World Medical Association Declaration of Helsinki. Written informed consent was obtained for publication of this case report and accompanying images and verbal informed consent was obtained for the other patients from themselves for their legal guardians.
A retrospective study of all patients who underwent free tissue transfers to the below-the-knee lower extremities due to chronic wounds during a 9-year period, between October 2011 and February 2020, was conducted. Patients with acute renal failure, undergoing hemodialysis or continuous renal replacement therapy, were excluded. ESRD was defined as renal failure that required regular dialysis.
Demographic data and medical history were obtained through retrospective chart review. The collected data included a detailed medical history, demographics, flap, and perioperative characteristics. Laboratory tests used for statistics such as serum creatinine, hemoglobin A1c (HbA1c), and C-reactive protein (CRP) were based on the first test results after admission to rule out effects from hydration or computed tomography (CT) contrast. The glomerular filtration rate was estimated using the Modification of Diet in Renal Disease equation (GFR=175×standardized serum creatinine−1.154×age−0.203×0.742 [if female]) [11]. Major complications were defined as those that required surgical intervention (exploration, debridement, repair, or additional coverage).
Statistical analyses were performed using IBM SPSS Statistics ver. 25.0 (IBM Corp., Armonk, NY, USA). Means and standard deviations were calculated for continuous variables and frequencies, and percentages were calculated for categorical variables. The t-test was used to examine whether there was a statistically significant difference in the means of the continuous variables between the two groups. The chi-square and Fisher exact tests were used to examine categorical variables. The logistic regression model for multivariate analysis was performed. A p-value of <0.05 was considered statistically significant.
Sixty-seven consecutive free flaps were used for chronic wounds of the lower extremity (below the knee) at our institution. The flaps included in the study were used in 52 male and 15 female patients. The mean patient age was 58.03±13.03 years (range, 29–82 years). The most common cause of defect was diabetic foot for 58 patients (Table 1). Fifty-three patients (79.1%) had diabetes; the average duration of their diabetes was 12.48 years, and their average level of serum HbA1c was 9.05% (Tables 2, 3). Three patients (4.5%) had a history of kidney transplantation. A total of 54 patients had PVD, and 41 (61.2%) underwent percutaneous transluminal angioplasty (Table 2).
The flaps used included the anterolateral thigh (ALT; n=57, 85.1%), medial sural artery perforator (n=7, 10.4%), vastus lateralis (n=2, 3%), and thoracodorsal artery perforator (n=1, 1.5%). Arterial anastomosis was performed by means of an end-to-side technique in 63 patients (94%). The most common site of coverage was the dorsum of the foot (n=18, 26.9%), followed by the forefoot (n=14, 20.9%), ankles (n=13, 19.4%), and heels (n=10, 14.9%). The procedures of 43 patients (64.2%) were performed on weight-bearing areas in the patients (Table 4).
As a result, flap failure occurred in six patients (9.0%), allowing us to obtain a total flap survival rate of 91.0%. Major complications occurred in 16 patients (23.9%) who required surgical intervention (Fig. 1, Table 5).
We performed a receiver operating characteristic (ROC) curve analysis to reveal the specific serum creatinine levels associated with the occurrence of flap failure. The sum of sensitivity and specificity was greatest when the serum creatinine level of 1.21 mg/dL and the area under curve of was 0.772 (Fig. 2).
The statistical analysis of patient characteristics revealed several statistically significant factors associated with major complications and flap failure after free flap surgery. In univariate analysis, flap failure rate was significantly higher in patients with ESRD and renal transplantation. Although not statistically significant, when serum creatinine levels were 1.21 mg/dL or higher, the p-value was 0.052, indicating a relatively high failure rate (Table 6).
Using multivariate analysis, ESRD and renal transplantation were associated with flap failure. The odds ratio was 77.8 and 86.7 respectively. Based on the serum creatinine 1.21 mg/dL obtained using the ROC curve, the analysis was conducted by dividing the group into a group with a serum creatinine lower than 1.21 mg/dL and a group with a serum creatinine higher than 1.21 but without dialysis. As a result, serum creatinine level was not related to flap failure (Table 7).
In the case of chronic wounds of the lower extremities, diabetic foot account for most cases, and PVD is often present, even without diabetes [12]. Therefore, we evaluated blood vessels of the lower extremity by performing CT angiography or conventional angiography in almost all patients. In addition, through a weekly conference with the radiology department, it was confirmed whether angioplasty would be beneficial to the patient, and if necessary, it was implemented. Most patients with chronic wounds often have wound infections, ranging from mild to severe infections. When angioplasty is required, it is usually performed, followed by wound preparation through debridement several times from the next day.
In the case of diabetic foot patients, many patients experience a long period of diabetes, and diseases such as coronary artery obstructive disease (CAOD) and CKD are often present. Free flap surgery is a lengthy procedure and sometimes results in significant bleeding. This may be particularly the case if patients are taking antiplatelet medications for CAOD or if they regularly use anticoagulants due to dialysis. Therefore, we consulted the Departments of Cardiology, Endocrinology, and Nephrology to confirm the necessary measures required before and after surgery. For ESRD patients, as much dialysis as possible should be performed the day before surgery, as a large amount of fluid enters during surgery.
We prefer donor sites with ipsilateral ALT, as ALT can obtain long and reliable pedicles. The flap was harvested from the ipsilateral side whenever possible to preserve the function of the contralateral leg as much as possible. For the donor site, vacuum-assisted closure is applied to decrease donor site complications and reduce the frequency of dressing changes.
In most cases, end-to-side anastomosis was performed to maintain the distal flow. In particular, patients with chronic wounds do not prefer end-to-end anastomoses because there are many patients with PVD, often comprising of patients with single-vessel lower extremities.
One of our postoperative management strategies is to have the patient sedated in an intensive care unit (ICU) for 24 to 72 hours after surgery. It is challenging to keep a patient with several comorbidities stable in an intubated state for a long time, but this has several advantages. By immobilizing the patient, the flap complications caused by patient movement can be reduced. Additionally, when emergency operations are required, surgery can be performed quickly. Arterial line monitoring can be performed in the ICU, and the patient’s blood pressure can be adjusted to high or low depending on the condition of the flap. In the case of ESRD patients, conventional hemodialysis is mostly performed in the ICU, but in some cases, continuous renal replacement therapy may be performed if blood pressure is low. The times at which to end patient sedation and extubation were determined by the surgeon after inspection of the flap. In most cases, extubation was performed between 24 and 48 hours.
The use of free flaps for chronic wound reconstruction is challenging in most cases [2]. However, it is important to note that Oh et al. [4] suggested that reconstruction of the diabetic foot through free flap surgery itself increases the survival rate of patients. In addition, Moran et al. [8] reported that free flap surgery could be sufficiently attempted in patients with renal disease through infection control through revascularization and serial debridement. Accordingly, free flap surgery in chronic wounds should be considered before amputation, if it is possible to salvage the limb.
Of the 67 patients included in this study, nine were ESRD patients, and three flap failures occurred, showing a 33% failure rate. It has a very high failure rate compared to previous studies, as Moran et al. [6] reported two flap failures in 33 patients with renal impairment or ESRD. Chien et al. [13] also reported one flap failure in 20 patients with ESRD. Nevertheless, in the study by Moran et al. [6], there were four additional cases of eventual amputation due to complications that occurred within one month after surgery, and Chien et al. [13] also showed that the final limb salvage rate was 80% due to other complications. As a result, the final limb salvage rate of patients with ESRD showed relatively similar results. Notably, wound healing was achieved without major complications in six patients, with the exception of three patients with primary flap failure among nine ESRD patients.
There are many causes of complications after free flap coverage in chronic wounds. In particular, patients with renal dysfunction have difficulties, such as vascular problems, delayed wound healing, bleeding tendency, and coverage of chronic wounds, which can pose great challenges to the reconstructive microsurgeon. Renal impairment decreases immunity by decreasing neutrophil function, impaired phagocytic function, natural killer cell activity, T- and B-lymphocyte function, and T-lymphocyte response to standard antigens [13-15]. According to previous studies, the correlation between factors that inhibit wound healing in patients with chronic renal impairment and free flap transfer and complications has been reported [6,12,13-17].
Although there are limitations due to the small sample size, flap failure occurred in two out of three renal transplantation patients and three out of nine ESRD patients. Due to the small sample size of renal transplantation and ESRD patients, there is a limitation in interpreting the results of multivariate analysis as it is. However, univariate analysis used for nonparametric test also showed a trend consistent with multivariate analysis. Although patients undergoing renal transplantation maintain relatively normal renal function, this phenomenon is thought to occur for two reasons. First, renal transplantation patients show a high change in the duration of diabetes mellitus (DM). Diabetic kidney disease is a cause of renal failure in the Republic of Korea. Most patients undergoing renal transplantation have severe diabetes, and DM vasculopathy in the lower extremities is often accompanied by long-term severe diabetes [18]. In patients with severe vasculopathy, the free flap is challenging, and the probability of failure is relatively high. A second cause of the high failure rate in renal transplantation patients is thought to be the immunosuppressants they were taking. The use of immunosuppressants makes wounds susceptible to infections [19-21]. In addition, patients who underwent a renal transplantation visit the hospital frequently; hospitalization is high, and the probability of infection with antibiotic-resistant bacteria in the wound is also relatively high.
Although not as much as renal transplant patients, most ESRD patients also have various comorbidities such as diabetes, so their immunity is impaired and microcirculation in the lower extremities is poor, so they may be vulnerable to wound infection of the lower extremities. Especially in ESRD patients, unlike renal transplant patients, the interstitial volume cannot be directly controlled, and this may affect flap failure in a different way than renal transplant patients. Free flaps take a long time and, in some cases, cause a lot of bleeding. In particular, in the case of ESRD patients, anticoagulation agents are periodically administered for dialysis, and antiplatelet drugs are sometimes taken for a long time in case of comorbidities such as CAOD or PVD. This can contribute to increased bleeding and leads to hypotension during surgery. For free flap surgery on the lower extremities, the use of peripheral vasoconstricting agents such as norepinephrine is discouraged. Although controversial, there is an opinion that perfusion to the flap can be reduced by vasoconstrictors. Therefore, to maintain blood pressure during free flap surgery, anesthesiologists try to maintain the effective volume of the patient by administering crystalloid. Patients with ESRD do not have the ability to directly excrete interstitial volume overload caused by crystalloid administration, which leads to tissue edema and increased tissue pressure. Therefore, it can lead to a decrease in microcirculation. Even if volume overload is controlled through dialysis after surgery, dialysis itself is a process that causes a decrease in the patient’s blood pressure and may also affect the microcirculation of the flap by reducing the perfusion pressure.
In addition, blood vessel in both ESRD and renal transplantation patients are usually accompanied by vascular calcification [20,21]. In particular, in the case of diabetes with ESRD or renal transplantation, chronic hyperglycemia with oxidative stress intensifies vascular calcification [22-24]. Microanastomosis in severely calcified vessels is technically difficult and risky. Although there was no calcification at the microanastomosis site, calcification at the proximal or distal area of recipient vessel may affect the blood flow to the flap. It is possible that this vascular calcification of ESRD and renal transplantation patients may have influenced the flap failure rate.
The significance of this study was to statistically examine factors that may be related to flap failure in the reconstruction of patients with chronic lower-extremity wounds. In addition, a wide spectrum was investigated from patients with normal renal function to ESRD. The results showed that the flap failure rate was significantly higher in patients with ESRD and renal transplantation.
Fig. 1.
Diabetic foot reconstruction with a free flap. A 48-year-old male patient with diabetic necrosis of the right foot. After serial debridement, the infection was controlled, and granulation tissue was observed. (A) Dorsal view and (B) plantar view after serial debridement. (C) Dorsal view and (D) plantar view two weeks after free flap coverage. (E) Wound revision was performed due to wound dehiscence in the weight-bearing area.

Fig. 2.
Receiver operating characteristic (ROC) curve for flap failure with serum creatinine levels. An ROC curve was drawn using patients’ serum creatinine levels. The area under the curve (AUC) was 0.772, and the cutoff value of serum creatinine to flap failure was 1.21 mg/dL.
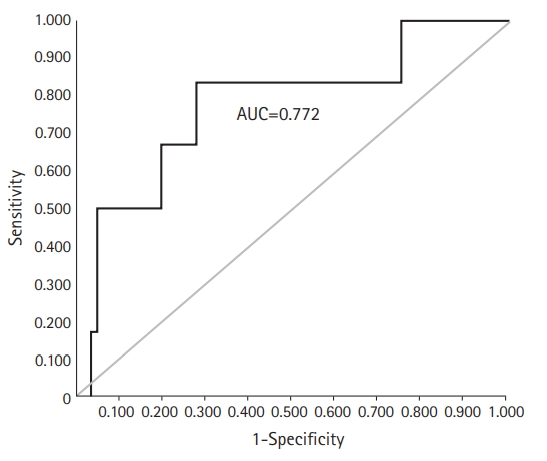
Table 1.
Patient demographics
Table 2.
Prevalence of comorbid illnesses
Table 3.
Preoperative laboratory test results
Table 4.
Flap characteristics
Table 5.
Postoperative complications
| Complication | Data |
|---|---|
| Total flap necrosis | 6 (9.0) |
| Partial flap necrosis | 8 (11.9) |
| Wound infection | 5 (7.5) |
| Wound dehiscence | 6 (9.0) |
| Microscopic exploration | 2 (3.0) |
Table 6.
Univariate analysis comparing the associations of various conditions with flap failure and major complications
| Major complication | No complications | p-value | Flap failure | Flap success | p-value | |
|---|---|---|---|---|---|---|
| End-stage renal disease | 3 | 6 | 0.437 | 3 | 6 | 0.028* |
| Renal transplantation | 2 | 1 | 0.139 | 2 | 1 | 0.020* |
| sCr level > 1.21 mg/dL | 7 | 13 | 0.218 | 4 | 16 | 0.052 |
| Recipient artery | 0.492 | >0.999 | ||||
| ATA | 14 | 38 | 5 | 47 | ||
| PTA | 2 | 13 | 1 | 14 | ||
| Angioplasty | 12 | 29 | 0.247 | 5 | 36 | 0.392 |
| Sex | 0.096 | 0.121 | ||||
| Male | 10 | 42 | 3 | 49 | ||
| Female | 6 | 9 | 3 | 12 | ||
| Weight-bearing reconstruction site | 5 | 19 | 0.662 | 1 | 23 | >0.999 |
Table 7.
Multivariate analysis of the associations of various conditions with flap failure
| Variable | OR (95% CI) | p-value |
|---|---|---|
| Duration of DM (yr) | 1.08 (0.95–1.22) | 0.240 |
| Renal transplantation | 86.75 (1.31–5,723.47) | 0.037* |
| sCr level | ||
| ≥1.21 mg/dLa) | 3.22 (0.05–207.74) | 0.581 |
| ESRD | 77.83 (1.18–5,100.53) | 0.041* |
| Angioplasty | 0.43 (0.02–10.24) | 0.605 |
References
1. Pu LL. A Comprehensive approach to lower extremity free-tissue transfer. Plast Reconstr Surg Glob Open. 2017;5:e1228.



2. Song JY, Kim KS, Kim HD, Park IS. Free tissue transfer in the treatment of infected diabetic foot ulcers. Arch Reconstr Microsurg. 2001;10:154-62.
3. Cooley BC, Hanel DP, Anderson RB, Foster MD, Gould JS. The influence of diabetes on free flap transfer: I. Flap survival and microvascular healing. Ann Plast Surg. 1992;29:58-64.


4. Oh TS, Lee HS, Hong JP. Diabetic foot reconstruction using free flaps increases 5-year-survival rate. J Plast Reconstr Aesthet Surg. 2013;66:243-50.


5. Ou CY, Wu MS, Lin MC, Chang CM. Short-term and long-term outcomes of free flap reconstruction versus amputation for diabetic foot reconstruction in patients with end-stage renal disease. J Plast Reconstr Aesthet Surg. 2022;75:2511-9.


6. Moran SL, Salgado CJ, Serletti JM. Free tissue transfer in patients with renal disease. Plast Reconstr Surg. 2004;113:2006-11.


7. Moran SL, Illig KA, Green RM, Serletti JM. Free-tissue transfer in patients with peripheral vascular disease: a 10-year experience. Plast Reconstr Surg. 2002;109:999-1006.


8. Illig KA, Moran S, Serletti J, et al. Combined free tissue transfer and infrainguinal bypass graft: an alternative to major amputation in selected patients. J Vasc Surg. 2001;33:17-23.


9. Attinger CE, Ducic I, Neville RF, Abbruzzese MR, Gomes M, Sidawy AN. The relative roles of aggressive wound care versus revascularization in salvage of the threatened lower extremity in the renal failure diabetic patient. Plast Reconstr Surg. 2002;109:1281-92.


10. Quiñones-Baldrich WJ, Kashyap VS, Taw MB, et al. Combined revascularization and microvascular free tissue transfer for limb salvage: a six-year experience. Ann Vasc Surg. 2000;14:99-104.


11. Levey AS, Coresh J, Greene T, et al. Using standardized serum creatinine values in the modification of diet in renal disease study equation for estimating glomerular filtration rate. Ann Intern Med. 2006;145:247-54.


12. Janhofer DE, Lakhiani C, Kim PJ, et al. The utility of preoperative arteriography for free flap planning in patients with chronic lower extremity wounds. Plast Reconstr Surg. 2019;143:604-13.


13. Chien SH, Huang CC, Hsu H, Chiu CM, Lin CM, Tsai JP. Free tissue transfers for limb salvage in patients with end-stage renal disease on dialysis. Plast Reconstr Surg. 2011;127:1222-8.


14. Haag-Weber M, Hörl WH. Uremia and infection: mechanisms of impaired cellular host defense. Nephron. 1993;63:125-31.



15. Lewis SL, Van Epps DE. Neutrophil and monocyte alterations in chronic dialysis patients. Am J Kidney Dis. 1987;9:381-95.


16. Mahmoodi BK, ten Kate MK, Waanders F, et al. High absolute risks and predictors of venous and arterial thromboembolic events in patients with nephrotic syndrome: results from a large retrospective cohort study. Circulation. 2008;117:224-30.


17. Yue DK, McLennan S, Marsh M, et al. Effects of experimental diabetes, uremia, and malnutrition on wound healing. Diabetes. 1987;36:295-9.


18. Kim B, Yoon J, Lim S, Han YS, Eo S. Anterolateral thigh free flap to cover diabetic foot defect by using reverse flow of severely calcified dorsalis pedis artery. Arch Hand Microsurg. 2021;26:293-7.


19. Sbitany H, Xu X, Hansen SL, Young DM, Hoffman WY. The effects of immunosuppressive medications on outcomes in microvascular free tissue transfer. Plast Reconstr Surg. 2014;133:552e-558e.


20. Lee AB, Dupin CL, Colen L, Jones NF, May JW, Chiu ES. Microvascular free tissue transfer in organ transplantation patients: is it safe? Plast Reconstr Surg. 2008;121:1986-92.


21. Åberg F, Isoniemi H, Höckerstedt K. Long-term results of liver transplantation. Scand J Surg. 2011;100:14-21.



22. D'Marco L, Bellasi A, Mazzaferro S, Raggi P. Vascular calcification, bone and mineral metabolism after kidney transplantation. World J Transplant. 2015;5:222-30.



-
METRICS

-
- 1 Crossref
- 1,978 View
- 41 Download
- Related articles in Arch Hand Microsurg
-
Sensory Function Recovery by Free Tissue Transfer in the Extremities2005 May;14(1)
Extensor Tendon Transfer for Chronic Rupture of Finger Extensors2012 June;17(2)






