Microsurgical Reconstruction of Lower Limb Using Thoracodorsal Artery Perforator Chimeric Free Flap after Popliteal Artery Revascularization: A Case Report
Article information
Abstract
Although traumatic popliteal artery injury is uncommon, it can significantly increase the risk of limb amputation because of the anatomical complexity and delayed diagnosis and treatment. Various tools are available for treatment. Recently, an endovascular approach has been attempted for such injuries; however, open surgical repair remains the standard treatment. An integrated and stepwise procedure involving multidisciplinary specialists, including emergency department personnel for initial evaluation, orthopedic surgeons for treating accompanying fractures or dislocations, vascular and plastic surgeons for vessel repair, and interventional radiologists for immediate diagnosis and implementation of the endovascular approach, is needed. Covering wound defects due to skin and soft tissue necrosis and irreversible ischemic damage remains difficult despite successfully revascularizing the injured vessels. Here, we describe a case of revascularization after popliteal artery injury along with successful reconstruction of a complex defect with a thoracodorsal artery perforator chimeric free flap when recipient vessel selection was limited.
INTRODUCTION
Acute popliteal artery injury with knee trauma is very rare, accounting for only approximately 0.2% of traumatic arterial injuries [1]. If the popliteal artery injury is not identified rapidly and managed appropriately, the prognosis of limb salvage is very poor, with a high risk of amputation [2]. Therefore, early diagnosis and prompt revascularization are crucial for limb salvage [3]. There are two approaches for rapid revascularization; endovascular and open surgery. Endovascular treatment includes removal of the thrombus, balloon angioplasty, and stent insertion. Open surgical repair involves a bypass using an autologous vein graft or allograft. Although a recent study reported similar outcomes for both methods, the standard of care remains to be open surgical repair, especially in patients with active external bleeding, expanding hematoma, exposed artery, or suspicion of complete vessel transection [1,4,5]. To this point, large-scale data on prognosis or long-term outcomes are lacking. Additionally, there are no data on microsurgical reconstruction using free tissue transfer after popliteal vessel revascularization. In this case report, we have shared our experience of microsurgical reconstruction using a thoracodorsal artery perforator (TDAP) chimeric flap for vessel-depleted limb salvage of popliteal artery obstruction and the complications after popliteal artery revascularization.
CASE REPORT
A 33-year-old male patient was referred to our unit after a motorcycle traffic accident. When the orthopedic surgeon initially assessed the patient’s condition, the blood vessels at the ankle were pulseless. The diagnostic impression was distal femur fracture with complex displacement, resulting in popliteal artery injury. Computed tomography angiography revealed a long segmental obstruction 8 cm above the tibial plateau (Fig. 1).

Posterior view of computed tomography angiography showing long segmental obstruction about 8 cm in length above the tibial plateau.
Emergency surgery was planned by the orthopedic, vascular, and plastic surgery teams. The orthopedic surgeon performed reduction and external fixation of the distal femur. Subsequently, the patient’s position was changed from supine to prone to facilitate a posterior approach for the popliteal artery injury. The vascular surgery and plastic surgery teams cooperated in repairing the popliteal vessel. Intraoperatively, the popliteal artery was found ruptured, and the length of the thrombus due to intimal injury was 10 cm. Therefore, thrombectomy and direct (proximal to distal) end-to-end anastomosis were deemed insufficient for revascularization. The injured artery was resected, and reconstruction method using allograft was chosen because it was difficult to harvest the autologous great saphenous veins. The popliteal artery was reconstructed by an allograft using the IMPRA Flex Graft (Impra Inc., Tempe, AZ, USA). Following reconstruction, the vascular flow, color of the lower limb, and pulse were restored. Intravenous heparin was infused immediately commenced, and blood flow was intact until 24 hours postoperatively.
On day 2 after the surgery, the pulses could not be detected by handheld Doppler at the ankle. Prompt angiography, performed in consultation with an interventional radiologist, revealed severe stenosis proximal to the anastomosis site. A thrombus was identified in the removed allograft during the emergency operation (Fig. 2) and popliteal artery reconstruction was performed by harvesting 10 cm of the autologous great saphenous vein from the ipsilateral donor site. Blood flow was intact intraoperatively. However, 24 hours postoperatively, although the popliteal pulse was intact, the distal pulse at the ankle level was weak. A radiologist performed angiography. Diffuse vasospasm was observed at the proximal level of the lower extremity in all vessels, including the anterior tibial, peroneal, and posterior tibial arteries, but there was no occlusion of the vascular anastomotic graft site (Fig. 3). Balloon angioplasty was performed, and a continuous vasodilator (prostaglandin E1) was administered via an intraarterial catheter. The patency of the posterior tibial and peroneal arteries did not improve due to vessel collapse. However, the blood flow in the anterior tibial artery was restored.
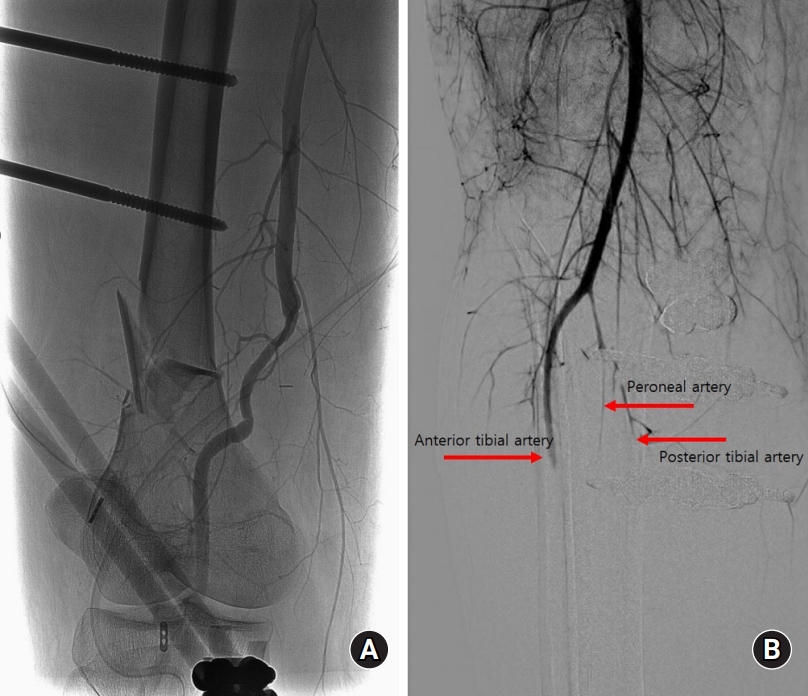
Angiography was performed by a radiologist. (A) No occlusions were observed at the anastomosis site. (B) Diffuse vasospasm was observed at the proximal level of the lower extremity in all vessels, including the anterior tibial artery, peroneal artery, and posterior tibial artery.
The orthopedic surgeon converted the external fixation to internal fixation 2 weeks after the trauma. The skin of the lower extremity was almost preserved, except for the heel and dorsal ankle area. Magnetic resonance imaging revealed necrosis and fatty degeneration of all muscles below the knee level, especially the gastrocnemius and soleus muscles (Fig. 4). We debrided necrotic tissue from the heel and posterior aspect of the calf, removing the severely necrotic portion of gastrocnemius and soleus muscle as much as possible. Not all necrotic muscles from ischemic damage developed wet gangrene with infection. The remaining ischemic muscles progressed to fibrosis. Even though these muscles had no remaining functions, they were preserved to maintain the shape of the leg and to spare the need for additional flap surgery.

Magnetic resonance imaging revealed diffuse muscle necrosis and fatty degeneration below the knee level, especially involving the gastrocnemius and soleus muscles.
The patient’s Achilles tendon was shortened due to contracture, and there were still soft tissue defects in the heel and dorsal area of the ankle. The orthopedic surgery team planned to release contracture and lengthen the Achilles tendon at an angle of 90°. Since the ankle was immobilized due to the surrounding tissue adhesion, no additional fixation procedure was required. The plastic surgery team contemplated covering multiple defects simultaneously. However, the patency of the patient’s vessels was not sufficient to perform multiple free tissue transfers to cover multiple defects (Fig. 5A–5C). Only the anterior tibial vessels were patent, while the remaining vessels below the knee level had collapsed. Consequently, we harvested a TDAP chimeric flap, which included 6 × 7 cm of latissimus dorsi muscle and a 16 × 12-cm skin perforator flap (Fig. 6). The skin flap was incorporated into the heel defect area, and the muscle flap was inserted into the dorsal ankle defect area with a split-thickness skin graft (Fig. 7). The thoracodorsal artery was anastomosed to the anterior tibial artery in an end-to-side fashion. The thoracodorsal vein was anastomosed to the vena comitantes of the anterior tibial artery in an end-to-end fashion.
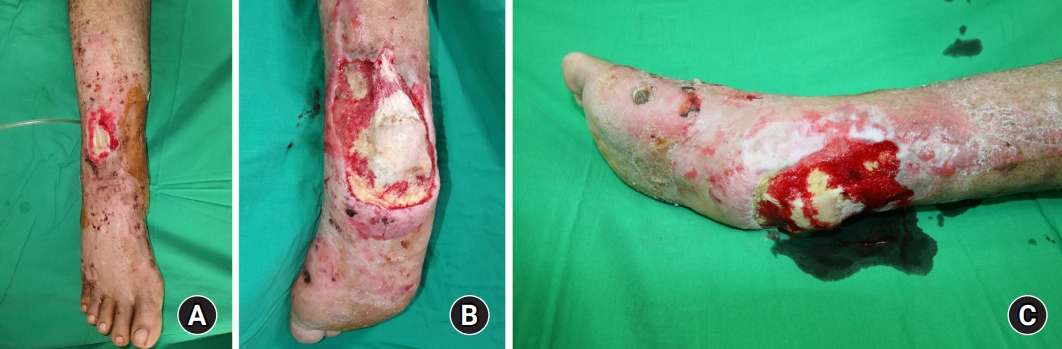
(A) Image showing the dorsal ankle defect with exposure of the extensor tendon. (B) Posterior view of the heel defect with exposure of the calcaneus and Achilles tendon. (C) Lateral view of the heel defect. This heel defect was managed with periodic negative pressure wound therapy, and the fresh granulation tissue had grown into the wound.
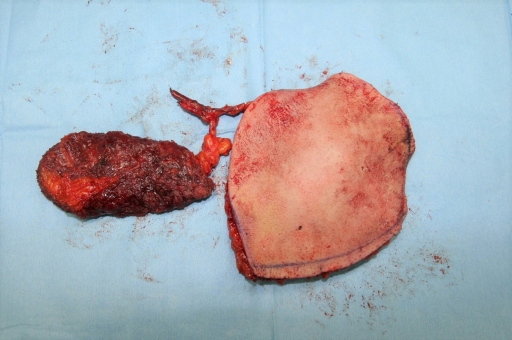
The harvested thoracodorsal artery perforator chimeric flap consisting of 6 × 7 cm-sized latissimus dorsi muscle and 16 × 12 cm-sized skin perforator flap.
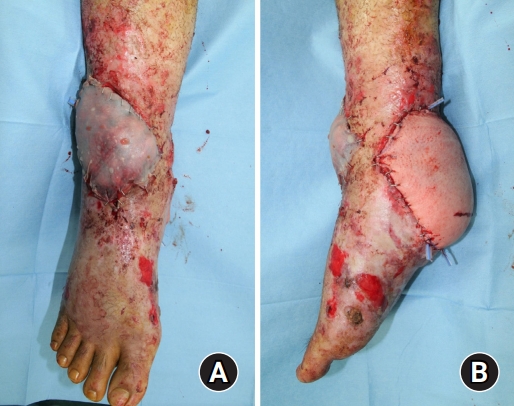
Immediate postoperative view. (A) A muscle flap was inserted into the dorsal ankle defect with a split-thickness skin graft. (B) Skin flap of the thoracodorsal artery perforator chimeric flap was incorporated into the heel defect area.
Most wounds healed without any complications besides calf area due to extensive calf muscle necrosis. Serial debridement was performed four times under local and general anesthesia. Finally, all the wounds were closed without any complications (Fig. 8). No wound problems were observed upon outpatient follow-up 7 months after injury. The patient could stand and walk without assistance. However, mild lymphedema was observed, and compression therapy was recommended.
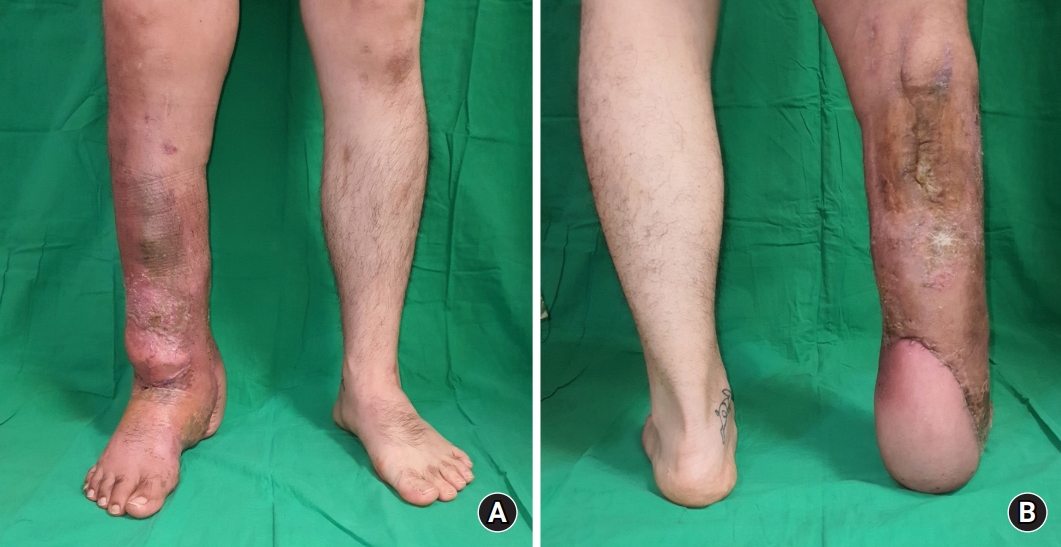
(A) Anterior view, 6 months after the trauma. (B) Posterior view, 6 months after the trauma. No wound problems were observed, and independent walking was possible.
Written informed consent was obtained for publication of this case report and accompanying images.
DISCUSSION
Popliteal artery injury due to trauma is rare. However, it can have potentially devastating consequences, with the amputation rate reaching as high as 14.5% in a recent study [2]. Popliteal artery injury is often accompanied by extensive soft tissue and bone damage around the knee joint, distal femur, and tibia. These vascular injuries are challenging owing to their complex management. The most important prognostic factor is the revascularization time from the injury. Exceeding revascularization time by 6–8 hours can result in an unacceptably high amputation rate [1,3]. Early intervention can help achieve superior outcomes and improve the chances of successful limb salvage [5]. Particularly, the skeletal muscle is vulnerable to ischemia, and critical tissue changes ensue if ischemia lasts for more than 4 hours. If the primary goal of popliteal artery revascularization has been successfully achieved, then functional recovery, such as independent walking, should be pursued. We have discussed and highlighted the lessons learned from this case.
First, the most important factor in treating popliteal artery injury is achieving revascularization as soon as possible [3]. In our patient, an orthopedic surgeon performed external fixation first for the distal femoral fracture. Subsequently, vascular reconstruction of the popliteal artery was initiated by the plastic surgery team after placing the patient in a prone position. In addition, the ruptured vessels and damaged surrounding tissues were intermingled, distorting the native anatomy; therefore, finding the target region required extra time. Surgeons should consider any means to reduce the revascularization time, such as using a temporary vascular shunt in complex extremity vascular injuries, which is known to reduce the ischemic time of the injured limbs and decrease the adverse effects of prolonged ischemia [6]. Accordingly, securing the circulation of the distal limb using a temporary vascular shunt before orthopedic surgery can be helpful.
The second lesson is that although prosthetic grafts have advantages such as easy availability, size matching, saving veins for subsequent revascularization, and reducing the operative time [7]. A meta-analysis by Sharrock et al. [8] showed that autologous grafts had superior patency rates and required fewer reinterventions. Therefore, an autologous vein graft should be considered first, and a prosthetic graft should be a subsequent option when an autologous vein graft cannot be used.
The third lesson is that an organized multidisciplinary team can reduce morbidity and increase the chances of limb salvage [5]. In our patient, the orthopedic surgeon helped achieve proper bone alignment, while the plastic and vascular surgeons cooperated, intraoperatively, to perform popliteal artery reconstruction. In addition, unnecessary invasive surgery was avoided, and complications like vascular spasms were promptly managed by early diagnosis and by using an endovascular approach such as intraarterial vasodilator infusion by the interventional radiologist [9].
The fourth lesson is that delayed thrombosis can occur following popliteal artery reconstruction. Therefore, the early detection of occlusion is vital. We checked the patient’s blood vascular status every 2 hours using a handheld Doppler ultrasound and pulsation at the ankle level, warmth, color, and capillary refill of the distal toes and foot. As a result, vascular problems were immediately detected and managed with surgical and endovascular approaches, leading to successful limb salvage in this case.
The final lesson is that TDAP chimeric flaps can be helpful in limited recipient vessel selection and complex wound coverage [10]. We used a TDAP chimeric flap to cover multiple wound defects. Although fresh granulation tissue was growing with periodic negative pressure wound therapy for the dorsal ankle defect, the extensor tendon and the surrounding soft tissue developed necrosis with inflammation. After the debridement, the wound defect and the dead space were enlarged and they were covered with TDAP chimeric free flap with good vascularity for wound healing. The muscle portion of the chimeric flap was harvested somewhat bulky to compensate for future muscle atrophy.
In this case, saving the limb was challenging because only the anterior tibial artery was intact below the knee area. Fortunately, the flap could be inserted very well, and all defects were closed without any infection or complications. Currently, the patient is able to walk without assistance and perform daily activities.
In this study, we dealt with long-term outcomes after successful revascularization and subsequent limb salvage. To the best of our knowledge, there are no cases that studied reconstruction of multiple complex defects followed by popliteal artery injury with TDAP chimeric free flaps. In addition, the blood vessels below the popliteal artery after successful revascularization could be used as a recipient vessel for free flap reconstruction of the lower extremity.
Notes
The authors have nothing to disclose.

